On October 8, 2022, the active biological bone products of Yantai Zhenghai Biotechnology Co., Ltd. were successfully approved for listing by the National Drug Administration (NMPA), marking the success of an epoch-making innovative medical device product designed and developed by Dai Jianwu, Institute of Genetic Development, Chinese Academy of Sciences, after 20 years.

Researcher Dai Jianwu was funded by the Hundred Talents Program of the Chinese Academy of Sciences in 2001 to establish a regenerative medicine laboratory in the Institute of Genetic Development of the Chinese Academy of Sciences. The scientific goal of the laboratory is to research and develop active biomaterials and products based on collagen biomaterials and tissue regeneration factors that can induce tissue and organ regeneration. At the beginning of its establishment, the active biological bone and spinal nerve regeneration scaffold will be the main core subject of the laboratory.
The goal of the team's initial design of active biological bone is to solve the side effects caused by excessive use of osteogenic growth factor (BMP2) in Medtronic active bone. Researcher Dai Jianwu believes that the biggest problem of Medtronic's products and later Stryker's active bone products is that the proliferation of osteogenic growth factor causes excessive dosage and obvious side effects. In order to solve this problem, the team designed the collagen-binding region obtained from collagenase and fused with BMP2 to obtain BMP2 that can specifically bind with collagen materials, named CBD-BMP2, and specially designed bone materials containing collagen. CBD-BMP2 combines with collagen-containing bone materials to form viable biological bone. Dr. Chen Bing, the first author of the research team, published a series of articles, proving that this original design can greatly reduce the use of BMP2 and obtain better osteogenic activity. As one of the first batch of graduate students recruited by Dai Jianwu Laboratory, Dr. Chen Bing is now an associate researcher of the Institute of Genetic Development, Chinese Academy of Sciences.
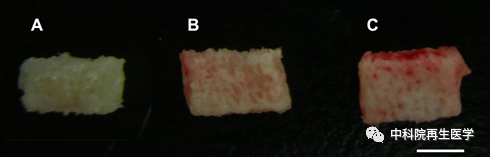
References above:
1.Chen, B., Lin, H., Wang, J., Zhao, Y., Wang, B., Zhao, W., Sun, W., Dai, J.* Homogeneous osteogenesis and bone regeneration by demineralized bone matrix loading with collagen-targeting bone morphogenetic protein-2. Biomaterials. 28(6):1027-35. 2007
2.Chen, B., Lin, H., Zhao, Y., Wang, B., Zhao, Y., Liu, Y., Liu, Z., Dai, J.* Activation of demineralized bone matrix by genetically engineered human bone morphogenetic protein-2 with a collagen binding domain derived from von Willebrand factor propolypeptide. J Biomed Mater Res A. 80(2):428-34. 2007
3.Zhao, Y., Chen, B., Lin, H., Sun, W., Zhao, W., Zhang, J., Dai, J.* The bone-derived collagen containing mineralized matrix for the loading of collagen-binding bone morphogenetic protein-2. J Biomed Mater Res A. 88(3):725-34. 2009
Another key technical problem in the research and development of active biological bone is the solubility of BMP2. The osteogenic growth factor BMP2 used in Medtronic active bone material can only be dissolved and stored in acidic solution, which is easy to cause calcium loss and is not conducive to bone regeneration. The protein purification team led by Hou Xianglin of Dai Jianwu's team and Chen Bing, an associate researcher, has established an original protein renaturation and purification process through nearly 10 years of efforts. The BMP2 prepared by the process can be dissolved in neutral solution, which makes the large-scale production of BMP2 possible. In 2011, Dai Jianwu's team completed the standardization research of CBD-BMP2.
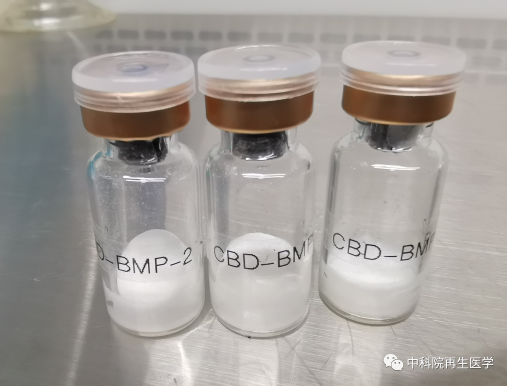
Figure CBD-BMP2 soluble in neutral solution
In 2015, Yantai Zhenghai Biological Co., Ltd. purchased the exclusive right to use the intellectual property rights of the active biological bone from the Institute of Genetic Development of the Chinese Academy of Sciences, and organized the multi-center clinical trial of the product. The company began to apply to NMPA for product registration in 2018. Since this product is a real original product with a breakthrough of 0 to 1, it was finally approved for registration and listing on October 8, 2022 after a long process of distribution and supplement. The approval of the active biological bone product marks that China's medical device innovation and evaluation has entered a new era. As the crownless king of today's medicated devices, the active biological bone will surpass similar products of Medtronic and Stryker from scientific basis to clinical application, marking that China's scientific research and innovation of medical devices has reached the international leading position.
Researcher Dai Jianwu proposed the concept of a human body 4S store based on regenerative medicine. His regenerative medicine innovation team has focused on the core technology innovation of regenerative medicine for 20 years, providing the underlying technology and products for human body 4S stores such as human tissue and organ regeneration care, repair and even organ manufacturing. In recent years, researcher Dai Jianwu has led more than ten clinical studies, including spinal cord injury regeneration, myocardial regeneration, endometrial regeneration, ovarian regeneration and lung regeneration, and has achieved encouraging results; Spinal cord regeneration, endometrial regeneration and ovarian regeneration were selected among the 40 achievements of the Chinese Academy of Sciences in the 40 years of reform and opening up; NeuroRegen stent, a regenerative repair product for spinal cord injury developed by the team, is undergoing multi-center clinical trials; The new generation of active biological bone developed by it is also about to be finalized. Dai Jianwu's team also hopes to cooperate with more enterprises and institutions interested in regenerative medicine innovation to jointly build a highland for innovation and transformation of regenerative medicine in China.